Clinical research is becoming increasingly data-intensive and operationally complex. Medical imaging is playing a growing role in evaluating treatment efficacy, monitoring safety, and supporting regulatory submissions across a wide range of therapeutic areas.
For Contract Research Organizations (CROs), imaging is no longer simply a supporting function. It is becoming a strategic operational capability that directly influences study timelines, data quality, and regulatory readiness.
Below are key trends expected to shape clinical trial imaging operations in 2026.
Clinical Trial Imaging Is Expanding Rapidly
Clinical trial imaging is experiencing steady growth as sponsors rely more heavily on imaging endpoints to measure treatment response and disease progression.
The global clinical trial imaging market, valued at approximately $1.42 billion in 2026, is on a high-growth trajectory toward $2.04 billion by 2031. This momentum is fueled by a fundamental shift in drug development: the move toward precision medicine and the integration of molecular-level visualization.
For CROs, this expansion signals a 2026 mandate for:
- Imaging Biomarker Specialization: Demand for imaging biomarker development services is growing at over 9% annually, as regulators increasingly require validated, quantitative endpoints.
- Centralized Orchestration: With Phase III trials accounting for over 40% of the imaging market, the ability to harmonize protocols across hundreds of global sites has become a core requirement.
- AI-Native Infrastructure: AI has moved from a “pilot” phase to a “production” phase, with nearly half of industry executives now rating AI-driven imaging as a top strategic driver for 2026.
CRO Outsourcing Continues to Increase
The global CRO market continues to expand as pharmaceutical and biotech companies outsource more clinical trial activities to specialized partners.
With the CRO industry valued at approximately $74 billion in 2026 and expected to grow significantly over the next decade, sponsors are increasingly relying on CROs for operational expertise, technology infrastructure, and specialized services such as imaging analysis.
As clinical trials grow more complex, imaging expertise is becoming an increasingly important capability CROs must provide in order to remain competitive and support sponsor expectations. This shift highlights a move toward strategic, long-term partnerships rather than the transactional vendor relationships of the past.
Imaging CRO Services Are a Fast-Growing Segment
Within the broader CRO industry, imaging services are emerging as one of the fastest-growing segments.
Demand for imaging CRO services is rising due to:
- the increasing use of imaging endpoints in clinical trials
- the need for centralized image review and adjudication
- regulatory expectations for standardized imaging workflows
As a result, the imaging CRO market is projected to approach $10 billion globally by the early 2030s, reflecting the growing reliance on specialized imaging expertise to support clinical development.
Why 2026 Represents a Tipping Point
The cumulative effect of regulatory clarity, AI maturity, workforce constraints, and operational complexity has elevated imaging infrastructure to a strategic consideration for CRO leadership. Sponsors increasingly assess not only scientific expertise, but also operational transparency and resilience.
CROs that treat clinical imaging as structured infrastructure, rather than a distributed vendor service, position themselves to protect margins, reduce timeline risk, and strengthen sponsor partnerships.
Those that continue to manage imaging through fragmented systems may find that inefficiencies compound as trial complexity increases.
The competitive distinction in 2026 is therefore structural, not incremental.
Clinical imaging is no longer simply about interpretation.It is about orchestration, control, and consistency.
And for CROs navigating increasingly complex development landscapes, that distinction matters.
AI and Cloud Technologies Are Transforming Imaging Workflows
New technologies are beginning to reshape how imaging data is managed in clinical trials.
Cloud-based imaging platforms enable secure sharing and centralized review of imaging data across multiple trial sites. At the same time, AI-assisted tools are helping improve workflow efficiency, triage imaging cases, and support quality control processes.
Together, these technologies are helping CROs manage increasing imaging data volumes while improving collaboration between investigators, radiologists, and sponsors.
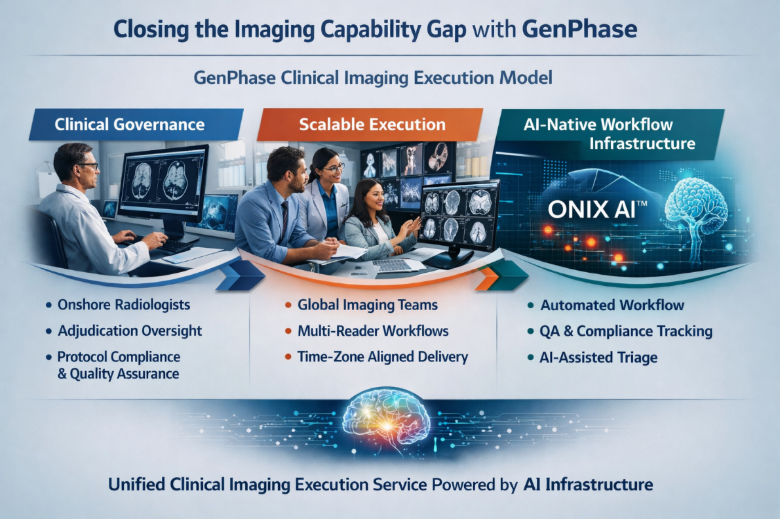
Closing the Imaging Capability Gap with GenPhase
As imaging requirements continue to grow in complexity, many CROs are exploring new service models that allow them to scale imaging operations while maintaining clinical rigor and regulatory alignment.
GenPhase provides end-to-end clinical imaging execution services for CROs and emerging biopharma sponsors, combining clinical expertise, scalable operational infrastructure, and AI-enabled workflow orchestration.
Clinically Led Execution with Global Scale
GenPhase’s model is built around onshore, board-certified clinicians who maintain full responsibility for imaging interpretation, adjudication, and reporting. This ensures consistent clinical oversight and regulatory alignment across the entire trial lifecycle.
Supporting this core is a credentialed global imaging team that enables scalable read capacity, faster turnaround times, and continuous coverage across time zones—allowing trials to operate without interruption.
AI-Native Workflow Orchestration
At the center of the delivery model is ONIX AI™, an AI-native orchestration layer that connects imaging data, clinical reviewers, and operational workflows into a unified system.
Rather than functioning as standalone software, ONIX AI™ enables:
- Protocol-driven workflow standardization across sites and studies
- Intelligent case routing and workload balancing
- Structured multi-reader and adjudication processes
- Integrated quality control and audit-ready compliance tracking
- AI-assisted triage to improve efficiency and throughput
A Unified Imaging Execution Model
By combining clinical leadership, global operational scale, and AI-driven orchestration, GenPhase enables CROs to:
- Scale imaging capacity without increasing operational complexity
- Improve turnaround time predictability across studies
- Maintain consistent, regulatory-aligned workflows
- Reduce fragmentation across imaging vendors and systems
This unified model allows CROs to scale imaging capacity, improve turnaround predictability, and maintain regulatory-aligned workflows, while reducing operational fragmentation across imaging vendors and systems.
Looking Ahead
Clinical trial imaging will continue to evolve alongside broader changes in drug development.
Increasing trial complexity, expanding imaging endpoints, and growing regulatory expectations are all driving the need for more robust imaging workflows and infrastructure. For CROs, imaging capabilities are becoming a key differentiator in delivering faster, more reliable clinical trials.
Organizations that invest in modern imaging systems, standardized workflows, and specialized expertise will be better positioned to support the next generation of clinical research.
Preparing for the Next Generation of Clinical Trials
As imaging becomes increasingly central to clinical trial execution, CROs and emerging biopharma sponsors must ensure their imaging operations are scalable, compliant, and operationally efficient.
GenPhase partners with CROs and emerging biopharma organizations to deliver end-to-end clinical imaging execution supported by AI-native workflow infrastructure.
By combining clinical expertise, scalable global imaging teams, and ONIX AI™ workflow orchestration, GenPhase helps organizations simplify imaging operations while maintaining regulatory-aligned delivery.
Schedule a strategy conversation to explore how GenPhase can support your clinical imaging programs.
Appendix: Sources
- Grand View Research — Clinical Trial Imaging Market Size, Share & Trends Analysis Report (2026-2030)
- Towards Healthcare — Clinical Trial Imaging Market Forecast: Impact of AI-Native Orchestration (2026 Edition)
- Allied Market Research — Clinical Trial Imaging Services Market Analysis: Trends in Oncology and Specialized Endpoints
- Coherent Market Insights — Imaging CRO Market Report: Projections through 2032 and the Rise of Radiomics
- Collective Minds — CRO Industry Market Statistics: The Shift from Outsourcing to Strategic Partnerships (2026)
- Precedence Research — Contract Research Organization (CRO) Market Size & Global Forecasts 2026
- FDA — Clinical Trial Imaging Endpoint Process Standards Guidance for Industry
- FDA — 21 CFR Part 11: Electronic Records and Electronic Signatures Guidance and Compliance Standards