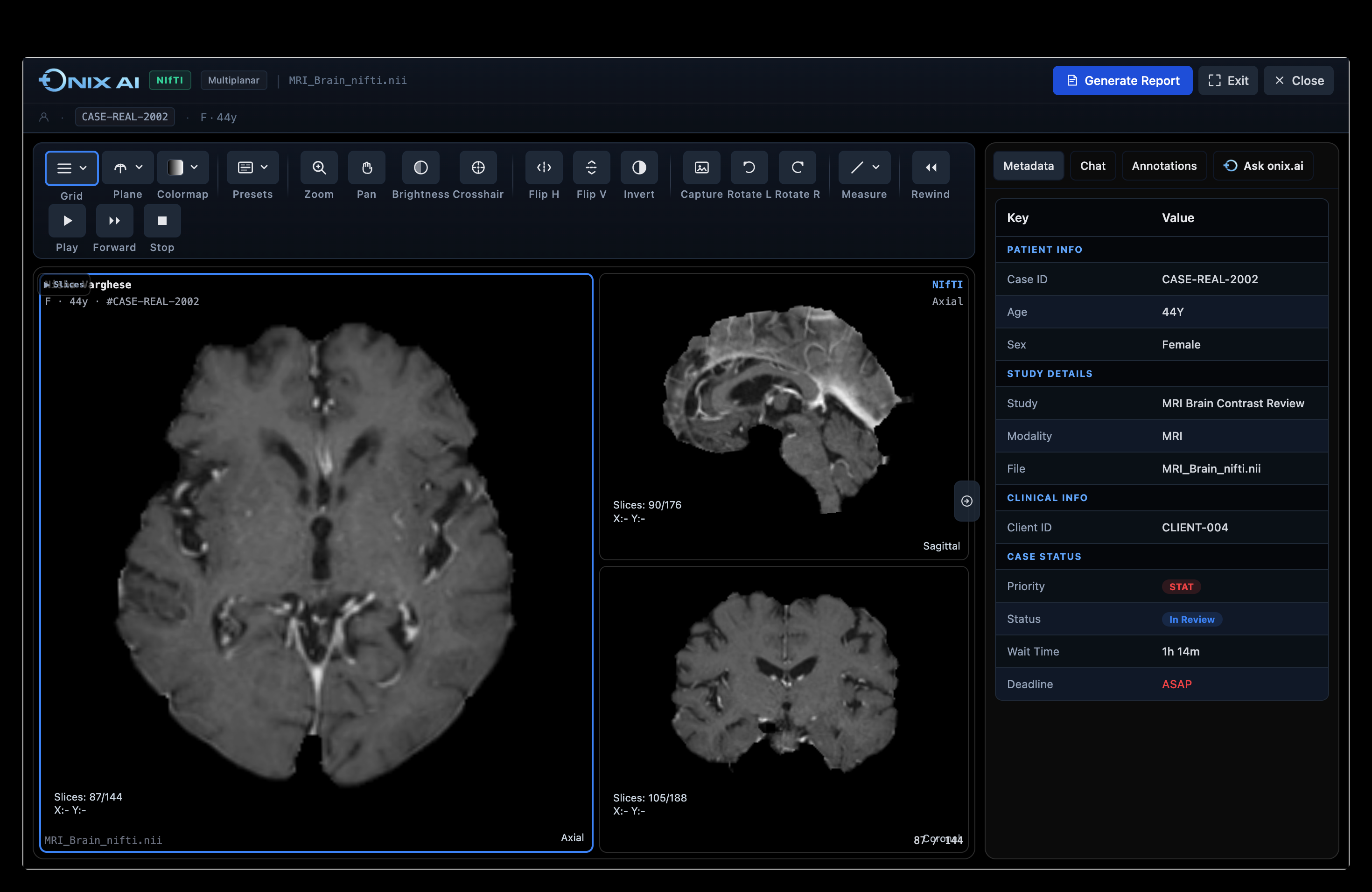
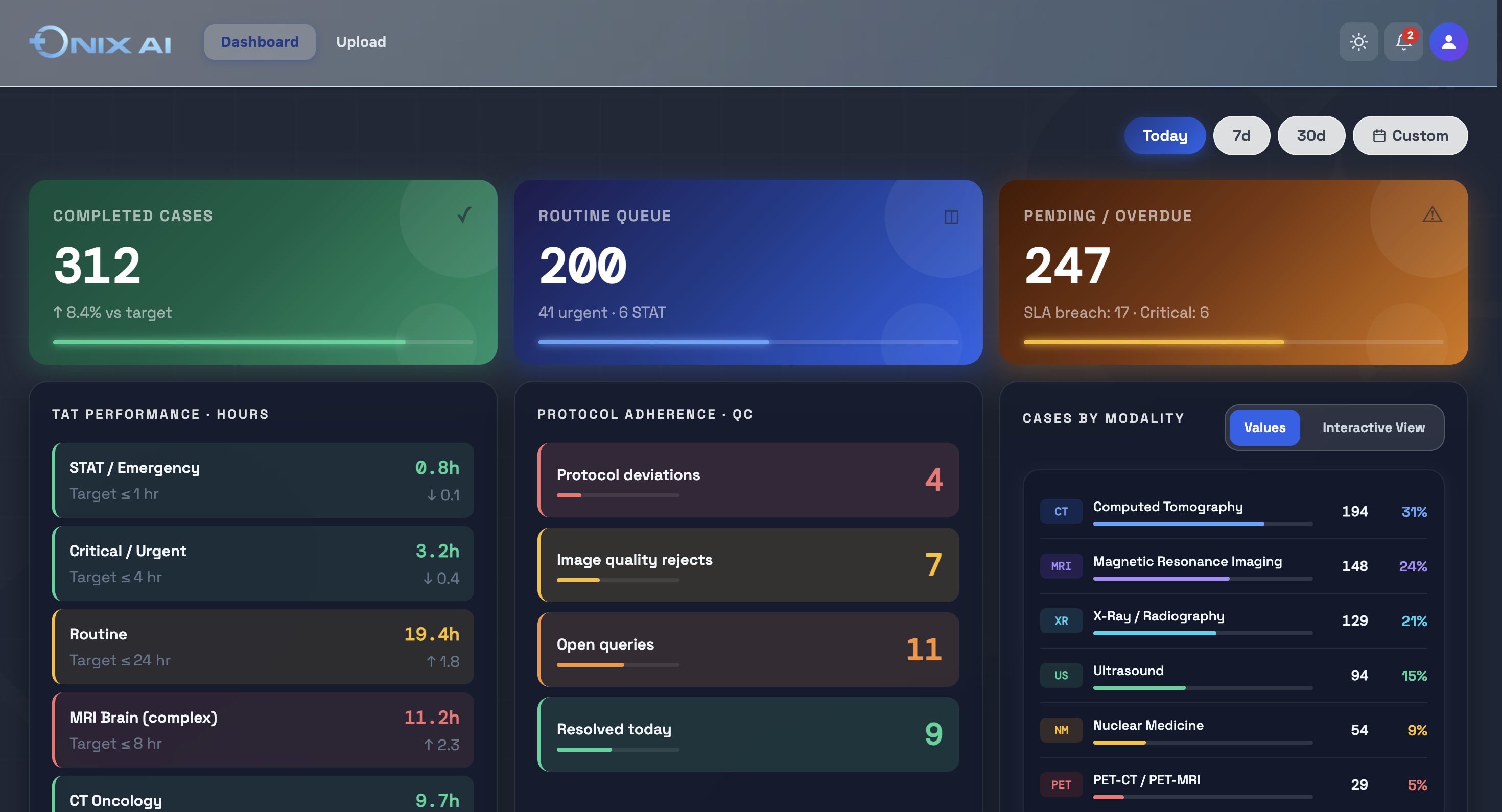
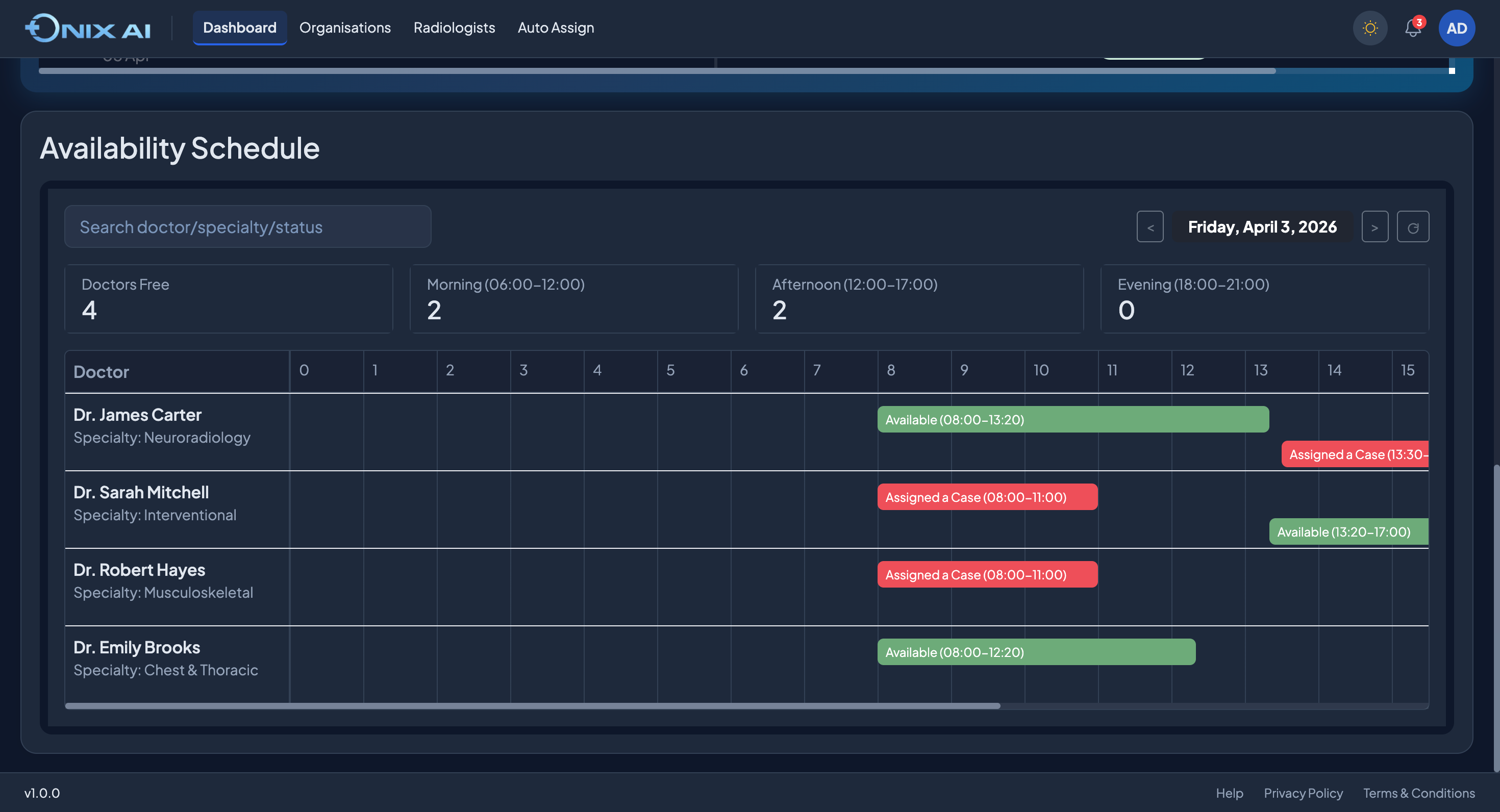
ONIX AI™ Clinical Imaging Orchestration.Built for Regulated Research.
ONIX is not an AI overlay on an existing PACS. It is a ground-up workflow engine designed for the specific compliance, data, and operational requirements of regulated clinical research.